Patient advocacy groups have urged the South African government to issue a compulsory license for a groundbreaking HIV prevention treatment after the Trump administration refused to include South Africa in a new program to distribute the drug to poor countries.
They argued the administration’s decision will not only harm efforts to thwart HIV in South Africa but also hinder a World Health Organization campaign to eradicate HIV by 2030. “No smart access effort can exclude South Africa from HIV prevention programming,” said Yvette Raphael, co-founder of Advocacy for Prevention of HIV and AIDS.
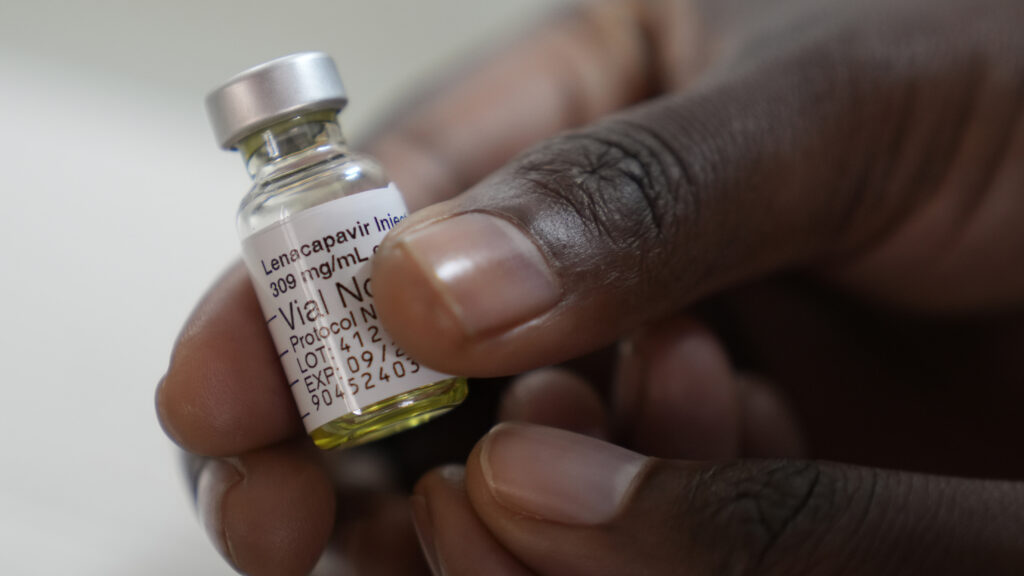
The appeal to South African officials comes after the Department of State earlier this week disclosed that 1,000 doses of the Gilead Sciences medication were recently delivered to Zambia and Eswatini, the first delivery under a plan announced late last year in which The Global Fund to Fight AIDS, Tuberculosis, and Malaria will distribute the drug.
Continue to STAT+ to read the full story…
STAT Pharma: The science and business of new drug development





Leave a Reply